Article: The Lotus Coffee Brew Water Kit: An Exploration of Dropper Variability

The Lotus Coffee Brew Water Kit: An Exploration of Dropper Variability
Written By: Nick Chapman
Introduction
The Lotus Coffee Brew Water Kit utilizes a system of concentrated mineral water and droppers to allow users to quickly and easily create water recipes or experiment with how different minerals can change our perception of the flavors present in coffee. I often get emails from customers regarding things such as how much a drop should weigh or how to verify that you are indeed creating the recipe that you intended to make. In answering these emails it has occurred to me that I have failed in some ways in our mission to make brewing great coffee accessible by not providing enough relevant information about the product and how it really works. This blog is an attempt to remedy that failure and will include a series of posts explaining exactly how The Lotus Coffee Brew Water Kit works, its limitations, and tips on how to get the most out of it. I hope you enjoy it.
How the dropper system works
Before we go any further though, it is necessary to briefly explain how a dropper system is supposed to work. In its simplest form, we are diluting concentrated mineral water of a known concentration in a known volume of distilled or reverse osmosis water to achieve a mineral concentration of our choosing. To correctly get to our desired end point we need to have control over three variables..
- The concentration of the mineral solution(s) to be diluted.
- The amount of water we are adding the mineral solutions to.
- The amount of mineral solution we are adding.
The first variable is easily solved by mixing the minerals and water in a large batch and doing so by weight. This is quite trivial to do accurately in a commercial manufacturing environment, and while slightly more challenging to do so in a home environment, with the right equipment and technique you can achieve similar results with relative ease as the accuracy of even the most basic scales is enough to achieve results with a margin of error below 1% as long as the batch you are making is large enough.
The amount of water we are adding to is also easy to solve for in both a home and commercial environment as the same principles discussed above also apply here.
The amount of mineral solution being added is where we run into some challenges. The easiest way to do this accurately is to make a less concentrated mineral solution and add significantly more concentrate than a dropper would. For example, you could create a concentrate such that adding 1 mL of concentrate to 1 L of water increases the concentration for that ingredient by 10 ppm as CaCO3. This approach works well for making your own water at home because even the most basic of scales are going to be able to deliver results with a reasonable level of accuracy.
The downsides here are that you need to make a significantly larger concentrate to make the same amount of water than if you made a much more concentrated version. For a commercial product, this means higher production cost, increased amount of packaging, increased shipping costs, etc. Additionally, you still need a way to consistently, conveniently, and accurately add 1 mL of concentrate to your water simple which usually involves a container to weigh out the concentrate and then transfer to your volume of water. Not exactly an elegant solution.
This is where we realize the beauty of using a dropper system. Surprisingly (or at least surprising to me), droppers are rather consistent in the amount of water they add per drop and that amount is rather small, less than a tenth of a gram. This allows us to make a much more concentrated solution and ditch the scale. I remember the first time I made a single dose batch of water using droppers and immediately realized that I could never go back to weighing out my concentrates again.
Dropper Accuracy and Precision
While using droppers solves many of our problems in crafting water, their effectiveness relies on their accuracy and precision, since we need to know exactly how much each drop is adding in order to accurately dilute our mineral solutions. To test this, I use a high precision scale that is calibrated and accurate down to the thousandths (0.001).
To collect the data presented below the following methodology was used.
Obtain one Lotus Brew Water Kit. Open each mineral concentrate. Dispense 10 drops into a dish placed on the scale, one drop at a time with the dropper held at a 15 degree angle off vertical. The weight of each drop is recorded and then the process is repeated for each dropper and each ingredient, performing the experiment twice and rinsing each dropper with distilled water before switching to a new ingredient.
This methodology produces data that allows us to see three things, the variability of drops for an individual dropper, the variability of drops between different droppers, and the variability of drops between ingredients.
Data and Analysis
If you don't like maths, feel free to just look a look at the graphs and then skip to the next section.

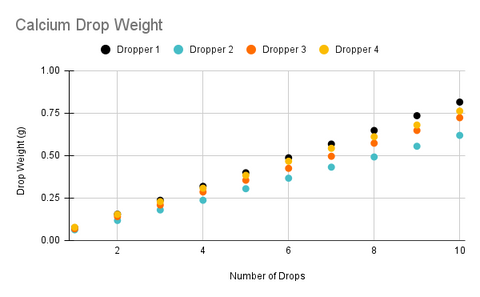
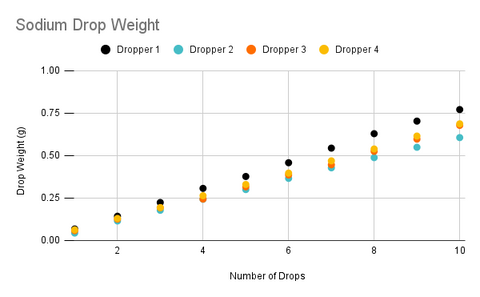
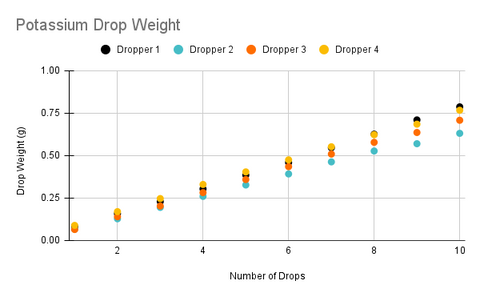
In the charts above, one thing that immediately stands out is that each dropper is dispensing a slightly different average drop weight with the relative standard deviation between droppers being right around 10% (If you are not familiar with relative standard deviation this means that 68% of drops from a randomly selected dropper will be within 10% of the average drop size of all droppers). Surprisingly, the variance between ingredients was not statistically significant as this was the case with the straight tipped droppers that were used in the original version of the product and one of the primary reasons we chose to switch to the new droppers as the previous droppers were dispensing almost a full 50% more for the calcium ingredient.
The data also reveals that each individual dropper also has a relative standard deviation of about 10%, however, the reason trend lines for each dropper look so consistent is because uncertainty decreases as we increase the number of drops. This because as we add more drops it is increasingly likely that the drops will cancel each other out and we will begin to approach the mean value. For example, if our first drop is 0.068g and our second drop is 0.077g the average drop size for those two drops is 0.0725, which is very close to the average drop size of all the droppers.
This is called the standard error of the mean (SEM) and can be calculated using the following equation.
SEM= s/√n
Where s is our standard deviation and n is the number of drops. Plugging in the data for dropper 1 we can see that the SEM for a 10 drop sample is:
SEM = 0.0059/√10 = 0.0018g
This means that if we are constructing a recipe that uses 10 drops of a single ingredient we will be within 5% of our target value 95% of the time.
Discussion
So what should we conclude from the results above? Well, it all depends on what you are trying to accomplish. I think an important follow-up study would be to attempt to find the flavor threshold for each ingredient as this would inform us of what level of accuracy is required to eliminate the ability to consider one recipe to be different from another. In other words, if you cannot tell the difference between two samples of coffee where the calcium hardness differs by 10 ppm from the other then there is no need to achieve an accuracy greater than +-5 ppm.
Another conclusion we can make is that if you are wanting to make the most accurate recipe possible you will want to increase the number of drops being added by mixing a larger volume of water. As a real world application of this and to see if the results above were indeed relevant to every day use I made a gallon of Light and Bright and tested the results. To do this, I calculated the SEM for this sample size and it came out to 3.3%. Then, I used a tool made by David Seng from The Espresso School based out of Melbourne to calculate a theoretical conductivity for this recipe which gave us 221.7 uS/cm. Lastly, I measured the resulting water with an HM Digital COM-80 handheld conductivity meter (TDS meter) that has a measurement accuracy of +-2% and was calibrated before the test using a sodium chloride solution. The meter read 219 uS/cm which serves as a pretty reasonable confirmation of our calculations.
One more thing to consider is that when you are making a smaller batch there is going to be an additional error from rounding since we cannot add anything other than a whole drop. When using the Lotus recipe calculator you may have noticed that changing the volume slider by a small amount does not always change the number of drops in the output table. This is because the calculator is targeting a specific ppm for that recipe but rounding the final value resulting in an additional error.
In practice, while knowing the exact composition of your water can be useful for communicating to others who may be using a different method of mineralization, I find it far more helpful to forget about ppm altogether and focus on dialing in the coffee by tasting it and simply adjusting the number of drops of each ingredient. As an example of this, whenever I get a new coffee, if it’s a washed process I will typically make a 450 mL batch of Light and Bright and taste the coffee. In this case, the easiest change to make is to adjust the number of drops of potassium to increase or decrease the perceived acidity. If the coffee was a little to acidic I will add another drop of potassium the next time. If I experienced the opposite I will use 1 drop less.
I hope this was helpful and that you are all having a great time brewing with Lotus. If you have any questions, comments, or thoughts on the above I would love to hear them. Just email me at Nick@lotuscoffeeproducts.com or reach out on Instagram.
**If you noticed any errors or misunderstandings in my presentation of the data I would love the opportunity to learn and you can send an email to nick@lotuscoffeeproducts.com explaining where you think the error was made and I will update this post accordingly

